 
If all three conditions are met then galvanic corrosion is probable and the rate of corrosion will be influenced by the relative area and the current density delivered by the noble metal. However, if the passive film breaks down, the stainless steel corrodes and its potential is in the dark bar range.Īs a rule of thumb, if the potential difference is less than 0.1 volt, then it is unlikely that galvanic corrosion will be significant. The usual, passive behaviour is shown by the light hatching. The graph shows that stainless steels have two ranges of potential. The rate of galvanic attack is governed by the size of the potential difference. The stainless steels, including 304 and 316, are more positive than zinc and steel, so when stainless steel is in contact with galvanised steel and is wet, the zinc will corrode first, followed by the steel, while the stainless steel will be protected by this galvanic activity and will not corrode. Zinc is more negative than steel and so the zinc coating of galvanised steel will corrode to protect the steel at scratches or cut edges. When two metals are connected and in contact with a conducting liquid, the more active metal will corrode and protect the noble metal. The potential can be measured with a reference electrode and is used to construct a galvanic series as shown below (ASTM Standard G82). 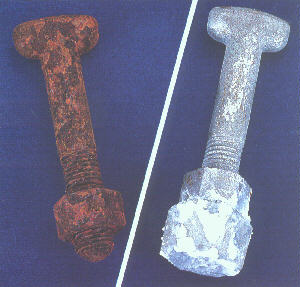
Stainless steel is in the middle although it is more noble than carbon steel. In contrast, noble or passive metals such as gold or graphite are relatively inert and have a more positive potential. The degree of dissolution is greatest with active or sacrificial metals such as magnesium and zinc and they have the most negative potential. Therefore the chromium oxide film will not prevent galvanic corrosion.Īll metals dissolve to some extent when they are wetted with a conductive liquid. 
Note that the chromium oxide film layer on the stainless steel is very thin and not an electrical insulator. The paint is usually damaged on installation or by subsequent movement. Paint is not a reliable electrical insulator especially under bolt heads or nuts or washers or near edges of sheets of metal. If the dissimilar metals are insulated from each other by suitable plastic strips, washers or sleeves then galvanic corrosion cannot occur. The contact may be direct or by an external pipe or wire or bolt. Galvanic corrosion can only occur if the dissimilar metals are in electrical contact. Painting the active metal (carbon steel or aluminium or zinc) can cause deep holes at coating defects. Water may be excluded by design or the use of adhesive sealants or by painting the noble metal for 30 to 50 mm beyond the join to prevent charged atom (ion) transport in any thin water film. One complication is that during evaporation, water films become more conductive so initially benign water may cause quite active galvanic effects as the liquid in the crevice under a bolt or clamp becomes more concentrated. Low conductivity, pure rainwater will only cause slight galvanic effects. 
Salt or industrial pollution significantly increases the conductivity of water so galvanic effects are normally more severe near the coast or in heavy industrial areas. The greater the conductivity the more severe the galvanic effects. If the deposits are sea salt, then they will start to dissolve if the RH exceeds 34% because of the magnesium chloride. The conductive liquid (or electrolyte) could be rainwater or water absorbed into surface deposits if the relative humidity (RH) is high enough or even simple condensation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
